Writing Clinical Research Protocols: Ethical Considerations: 9780122107511: Medicine & Health Science Books @ Amazon.com

TSDP explains the key-points of a #clinical study protocol under its #regulatory medical writing training program. | Writing lessons, Writing, Writing services

Medical Writing for Essential Clinical Trial Documents: A training manual to learn & create clinical trial documents: 9781511451437: Medicine & Health Science Books @ Amazon.com
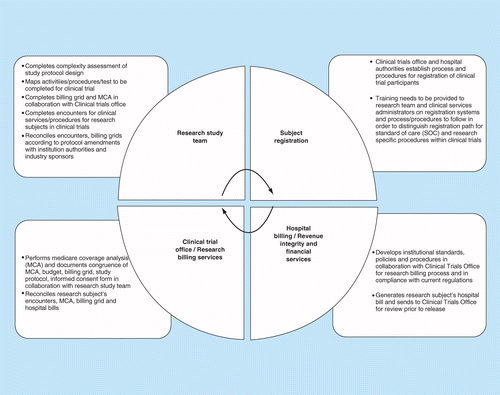
Optimization of protocol design: a path to efficient, lower cost clinical trial execution | Future Science OA

Writing Clinical Research Protocols: Ethical Considerations: 9780122107511: Medicine & Health Science Books @ Amazon.com