Combination of Atezolizumab and Tazemetostat in Patients With Relapsed/Refractory Diffuse Large B-Cell Lymphoma: Results From a Phase Ib Study - ScienceDirect

Cancers | Free Full-Text | Evaluation of Tazemetostat as a Therapeutically Relevant Substance in Biliary Tract Cancer

EZH2 inhibitor tazemetostat in patients with relapsed or refractory, BAP1-inactivated malignant pleural mesothelioma: a multicentre, open-label, phase 2 study - The Lancet Oncology

Testing the Addition of Tazemetostat to the Immunotherapy Drug, Pembrolizumab (MK-3475), in Advanced Urothelial Carcinoma
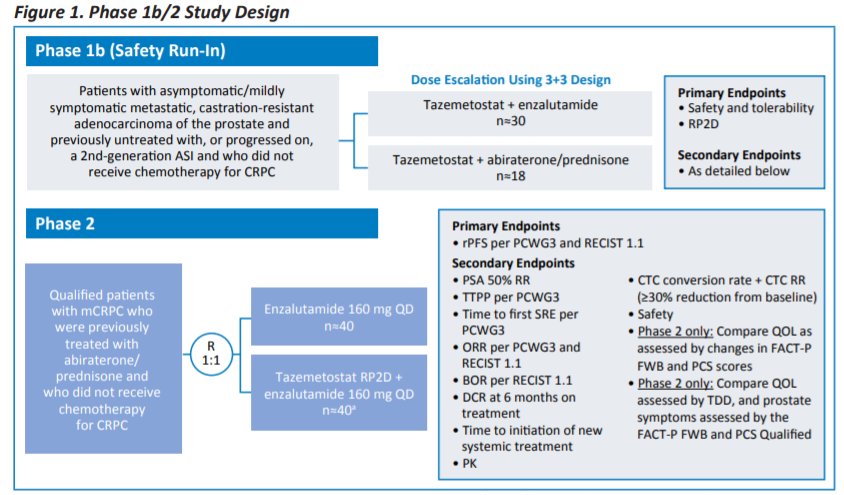
ESMO 2021: Safety of Tazemetostat in Combination With Abiraterone/Prednisone or Enzalutamide in Patients With Metastatic Castration-Resistant Prostate Cancer

Discovery of Dual Lysine Methyltransferase G9a and EZH2 Inhibitors with In Vivo Efficacy against Malignant Rhabdoid Tumor | Journal of Medicinal Chemistry

The EZH2 inhibitor tazemetostat upregulates the expression of CCL17/TARC in B‐cell lymphoma and enhances T‐cell recruitment - Yuan - 2021 - Cancer Science - Wiley Online Library

Discovery of a Novel Covalent EZH2 Inhibitor Based on Tazemetostat Scaffold for the Treatment of Ovarian Cancer | Journal of Medicinal Chemistry

Epizyme Announces U.S. FDA Accelerated Approval of TAZVERIK™ (tazemetostat) for the Treatment of Patients with Epithelioid Sarcoma | Business Wire

Tazemetostat for patients with relapsed or refractory follicular lymphoma: an open-label, single-arm, multicentre, phase 2 trial - The Lancet Oncology

OncLive.com on Twitter: "Watch our upcoming live broadcast reviewing a new indication, clinical trial data, and dosing for TAZVERIK TM (tazemetostat): https://t.co/YKnSN20tj9 https://t.co/ZfU6XQc4Gd" / Twitter

Epizyme Announces the U.S. Food and Drug Administration Lifts Partial Clinical Hold on Tazemetostat Clinical Program | Business Wire

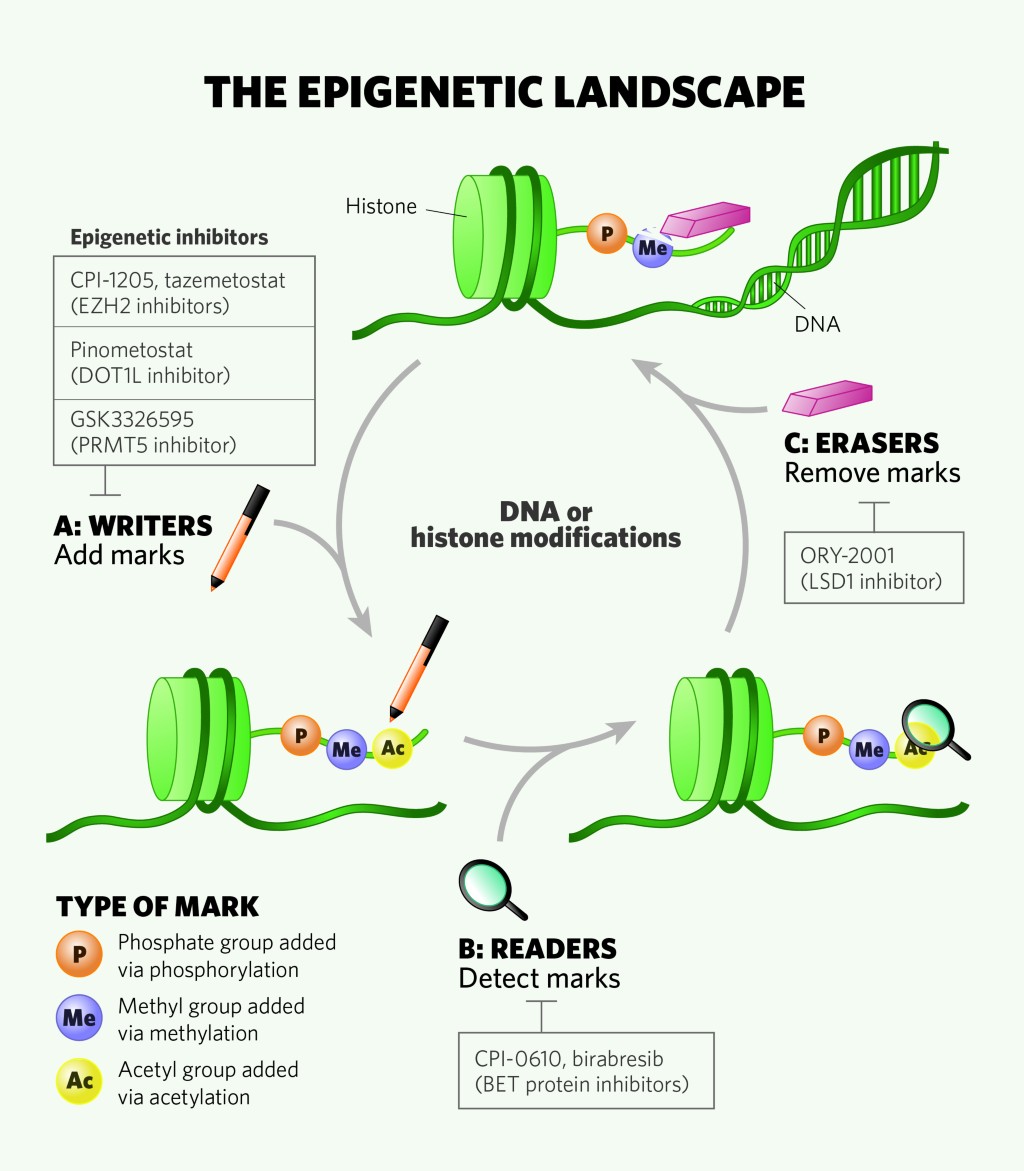
Targeting Enhancer of Zeste Homolog 2 for the Treatment of Hematological Malignancies and Solid Tumors: Candidate Structure–Activity Relationships Insights and Evolution Prospects | Journal of Medicinal Chemistry