Teva Accused Of Minimizing Its Part In Opioid Epidemic | MedTruth - Prescription Drug & Medical Device Safety | Informed Advocacy

Teva completes $703M sale of women's health products to CVC Capital Partners. - Philadelphia Business Journal
Teva Ketoconazole Cream 2% Topical Antifungal Cream 30 gram tube (Rx) — Mountainside Medical Equipment

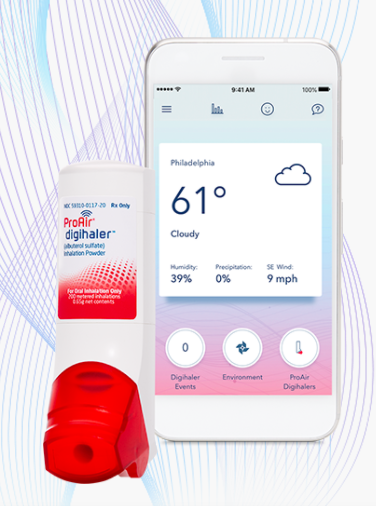
Teva Launches Two Digital Inhalers in the U.S., AirDuo® Digihaler® (fluticasone propionate and salmeterol) Inhalation Powder and ArmonAir® Digihaler® (fluticasone propionate) Inhalation Powder

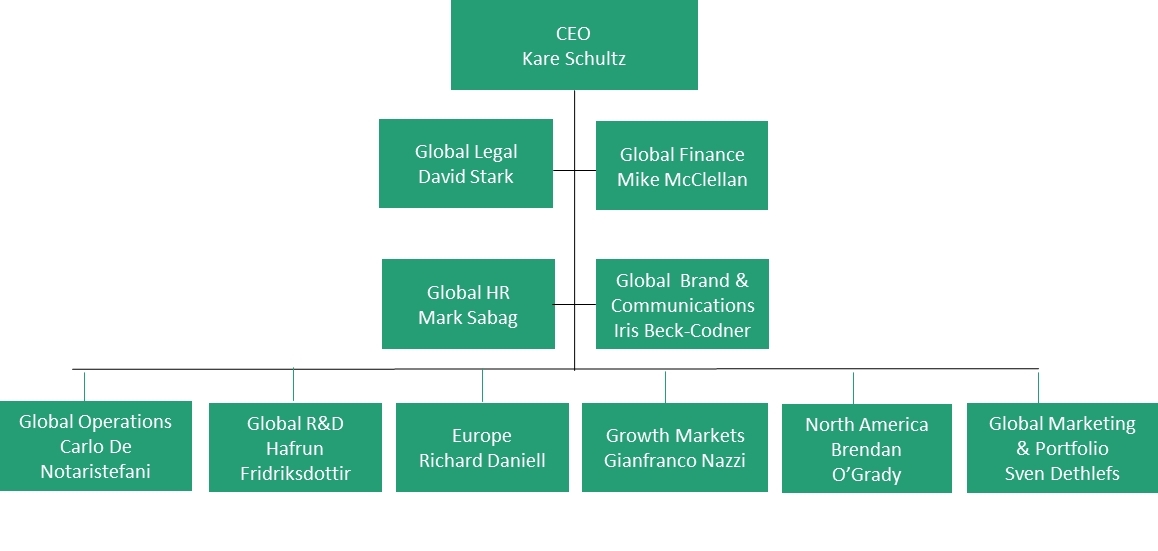
Melissa Kendall - Sr. Director Combination Products and Devices R&D - Teva Pharmaceuticals | LinkedIn