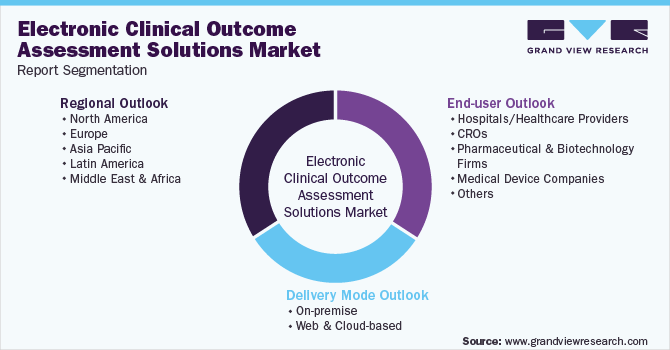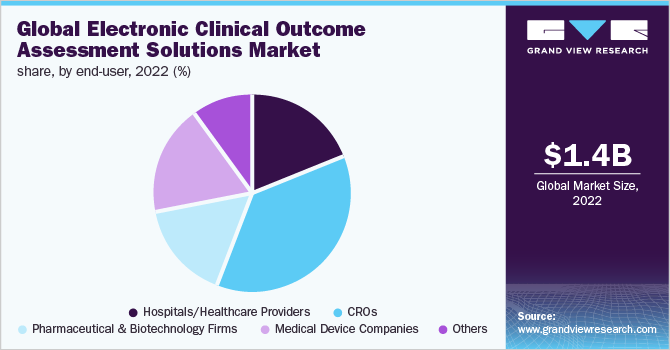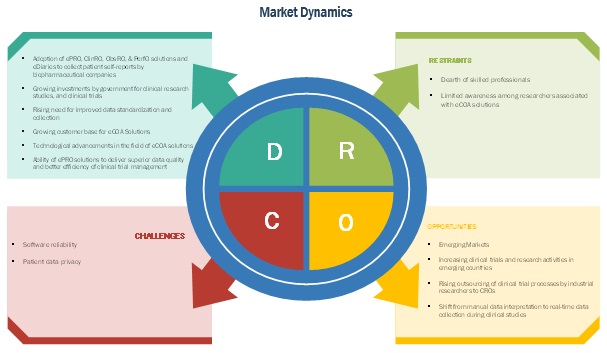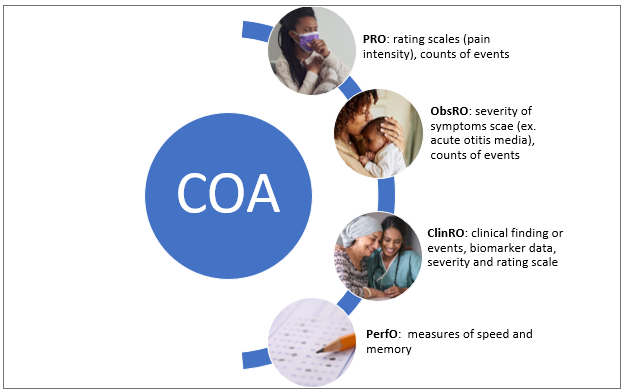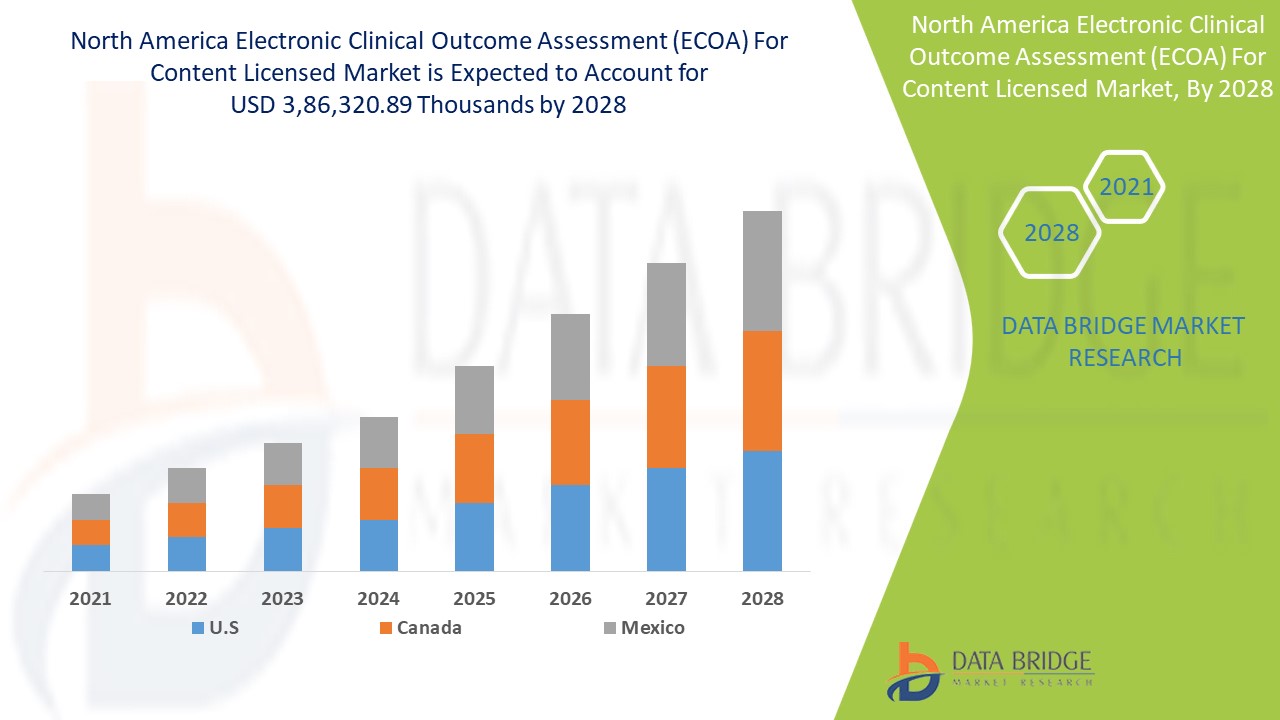
North America Electronic Clinical Outcome Assessment (eCOA) for Content Licensed Market Report – Industry Trends and Forecast to 2028 | Data Bridge Market Research
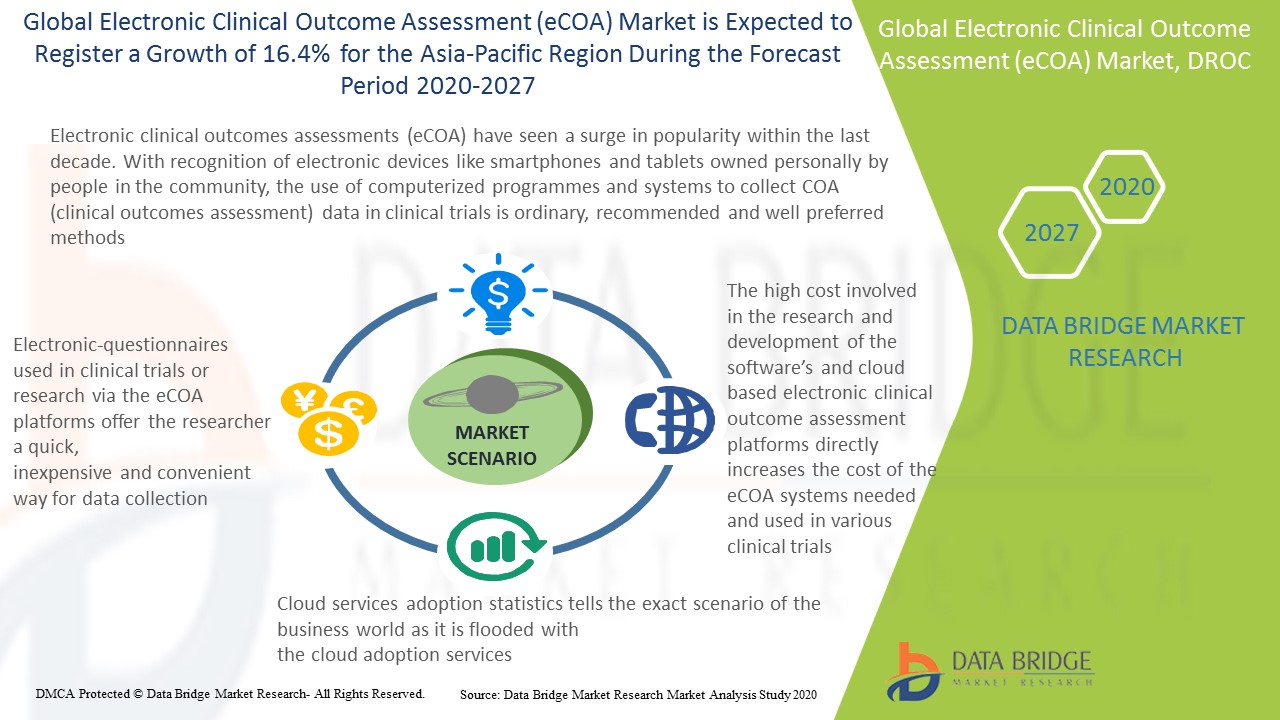
Global Electronic Clinical Outcome Assessment Ecoa Market Research Report, Future Demand and Growth Scenario

Clinical Outcome Assessments: Accurately Reporting Measurements in any Language - Welocalize Life Sciences

Towards a novel clinical outcome assessment for systemic lupus erythematosus: first outcomes of an international taskforce | Nature Reviews Rheumatology
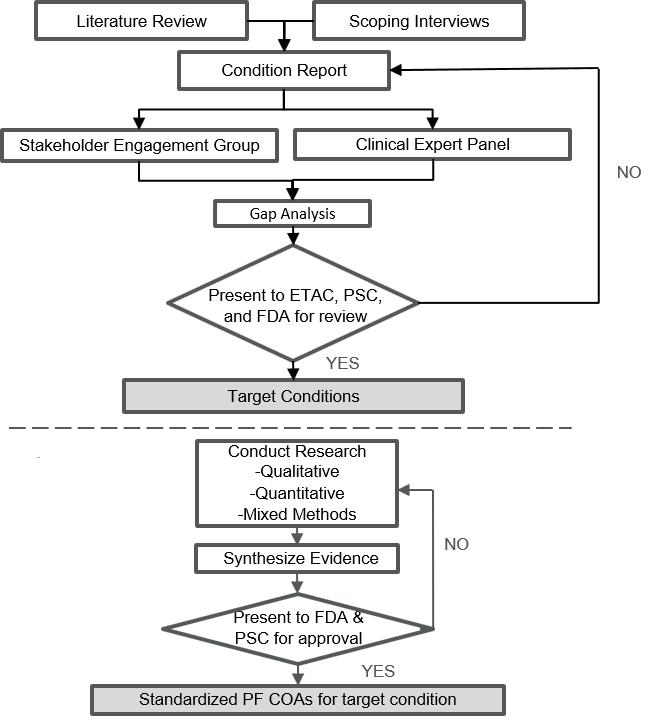
This Paper focuses on CDISC Questionnaires, Ratings and Scales (QRS) supplements and types of FDA Clinical Outcome Assessments