The ethics of conducting clinical trials in the search for treatments and vaccines against COVID-19 - FPM

Vulnerable population and methods for their safeguard – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka open science hub.
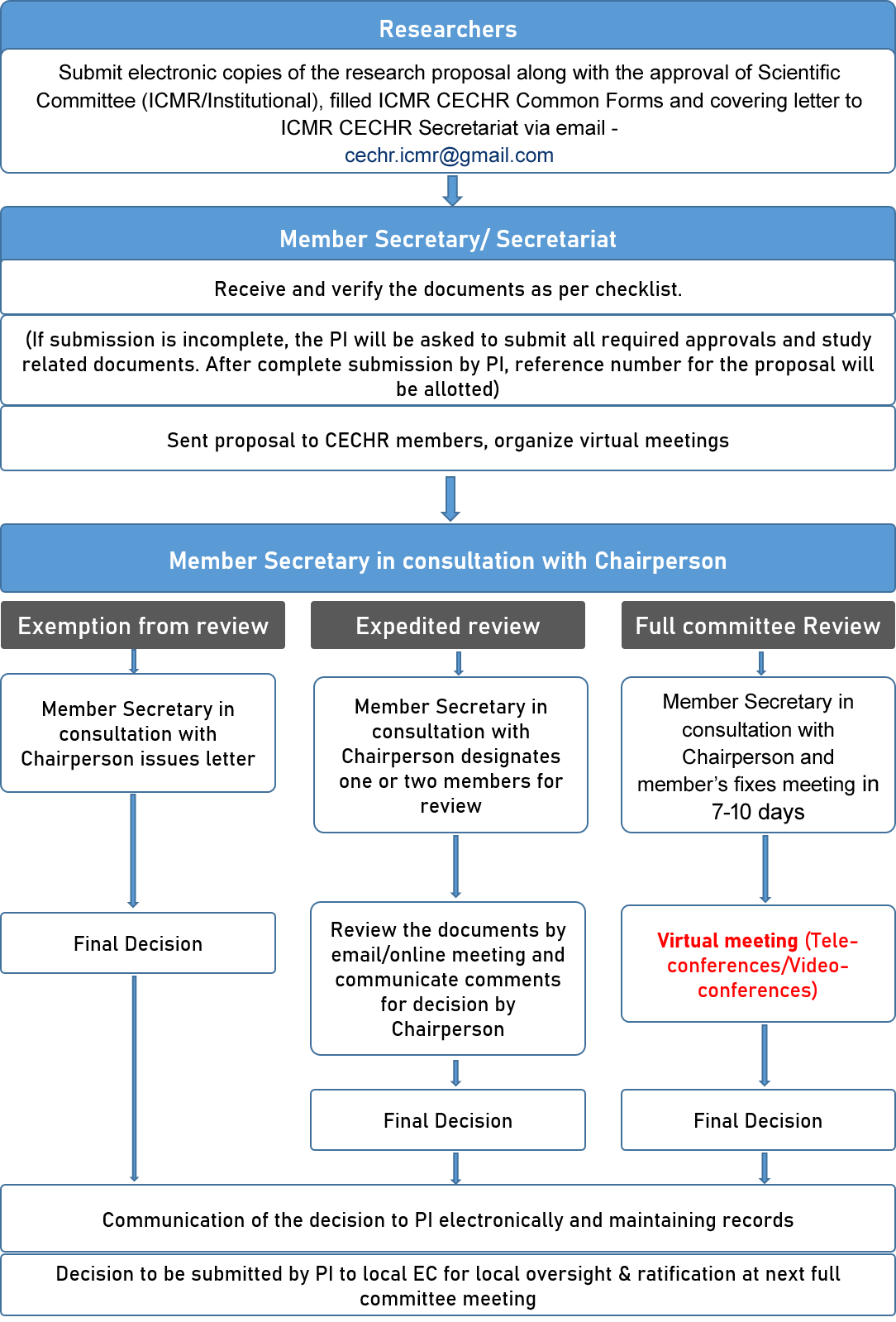
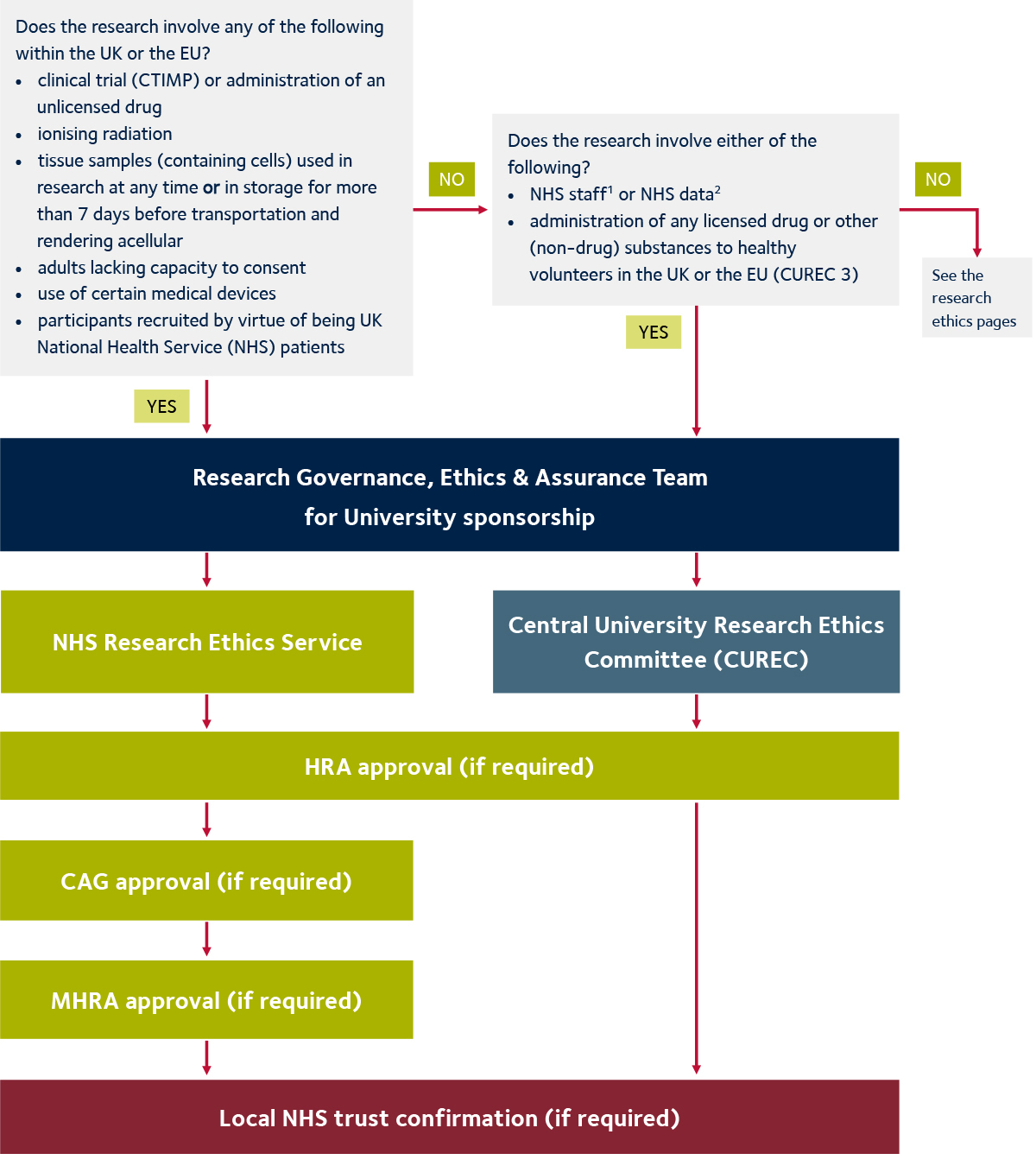
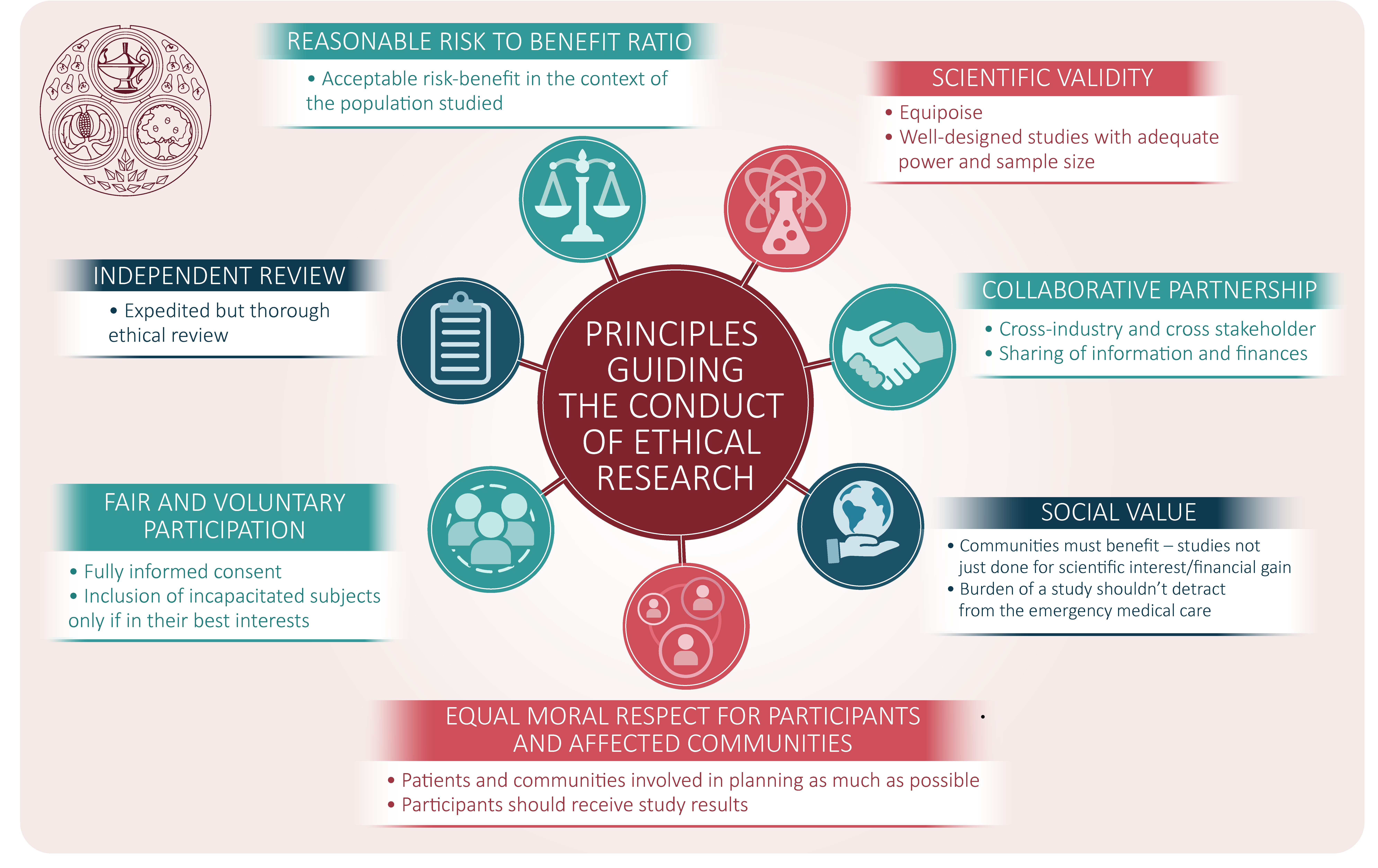
Guidance for research ethics committees for rapid review of research during public health emergencies

Clinical Trial Clinical Research Ethics Committee Therapy, PNG, 1679x1096px, Clinical Trial, Adverse Effect, Cartoon, Clinical Research,

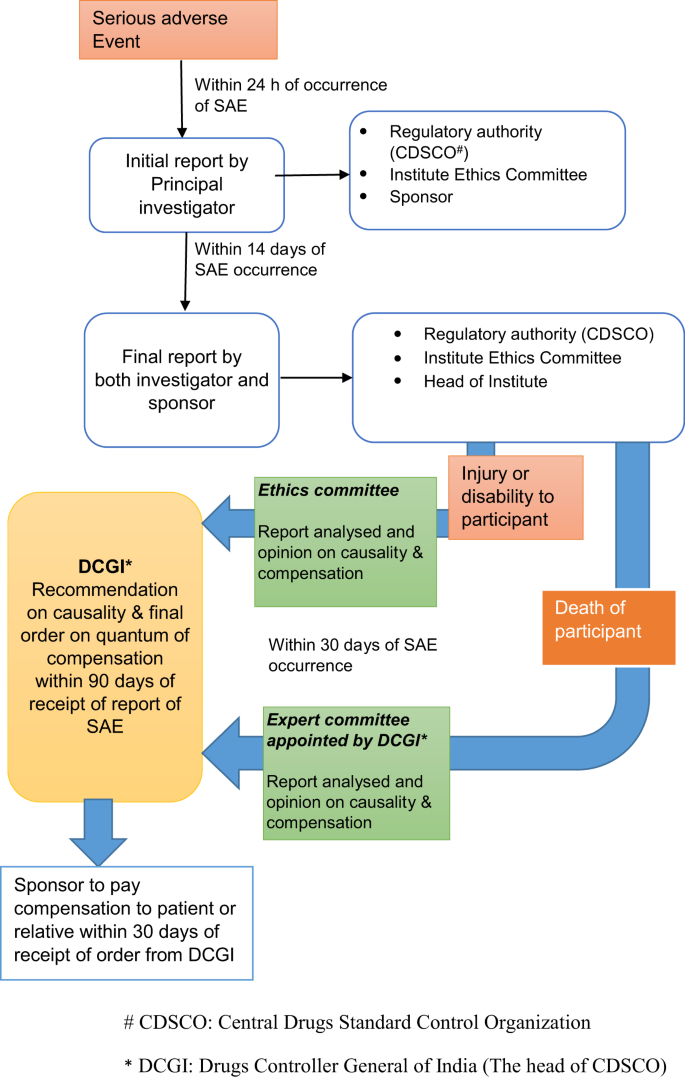
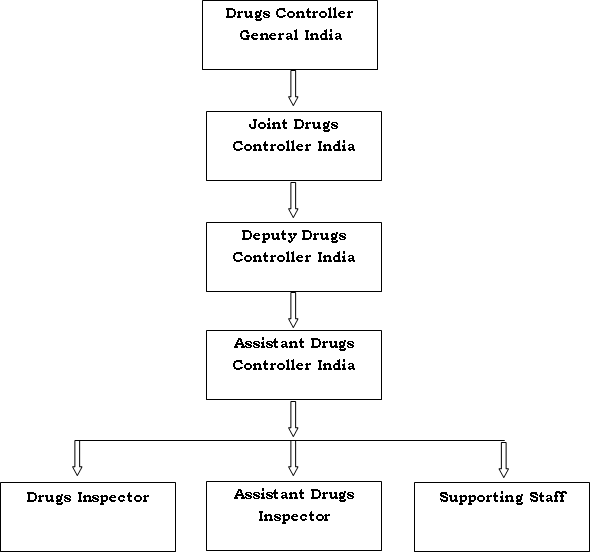
Oct 06 Ethical and Regulatory Approval Process in HIV Vaccine Clinical Research India Experience HIV Vaccine Clinical Research Ethical and Regulatory Issues. - ppt download